Addictive Bacterial control system- TAT
Greetings
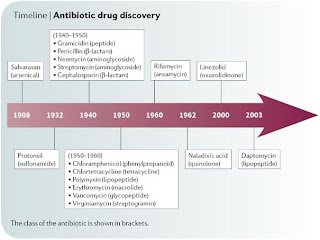 |
| Fig 1: Antibiotic discovery timeline. Source |
One of the first few posts, when I started blogging, was to write about antibiotics. I talked about use of halocins, targeting quorum sensing, attacking the cell replication machinery and toxin antitoxin system. Considering the field of infectious diseases a lot of study goes into studying antibiogram and spread of antibiotic resistance in several species. With more and more organism raising to the status of MDR (multi-drug resistance), XDR (Extremely drug resistant) and superbugs, we are short of antibiotics. To cite an example, MRSA (Methicillin resistant Staphylococcus aureus) is treated with vancomycin. resistance to vancomycin leaves the clinicians with almost no choice of drugs. Vancomycin was discovered as early as 1960's. The latest class of drugs like carbapenems and puromycins was discovered in 1980's. Practically, we have not discovered an antibiotic for more than 30 years (Daptomycin is an exclusion)!!!
 |
| Fig 2: Mechanism of toxin antitoxin action. |
Now let me shift my focus of this blog post- Toxin antitoxin system. The system is a bi-component system. The two components include a toxin that has a long half life, and an antitoxin with a short half life. The point is the the toxin will be produced by the bacteria and is lethal to itself, until there is a antitoxin expressed somewhere in the genome to save itself. This provides a strong control over a variety of factors. The essence of this mechanism is most appreciated in maintenance of plasmids, population control etc. For example, the genome encodes toxin and plasmid encodes antitoxin. If plasmid is lost cell is dead.
As can be seen there is a great deal of opportunity to create an antibiotic here. The point is the mechanism is excruciatingly simple. But there are a variety of molecules and operons involved in a singles system. If i remember it correct, a single bacterial cell can possess several sets of such systems. Based on the molecules involved, the mechanism of action, Toxin antitoxin (TAT) has been classified into 5 different class.
 |
| Table 1: Classification of Toxin-Antitoxin system (TAT). |
With this background information and understanding, lets take a look at one example of how the system functions. For elaboration purpose, am citing the MazEF system of E coli. It is one of the best studied with sufficient data available to see the point.
The MazEF system consists of MazF that encodes a stable toxin and MazE encoding a labile antitoxin which prevents the lethal effect of MazF. MazF has a sequence-specific ribonuclease activity toward single- or double-stranded RNA regions and the resulting degradation of cellular mRNA causes global translation inhibition. To be precise, action of MazF is to cleave at ACA site closely upstream of the AUG start codon of some specific mRNAs and thereby generating leaderless mRNAs. A true toxin indeed. The system is often referred as Programmed Cell Death (PCD), analogous to apoptosis in eukaryotic cells. Since the cells cannot be sustained without the antitoxin, the antitoxin is vaguely referred as "Addiction molecule".
 |
| Fig 3: MazEF system. |
The MazEF system is linked to the quorum sensing system. Triggering of extra cellular death factor (EDF), which is a quorum-sensing pentapeptide NNWNN leads to cascade of events. zwf gene product (glucose-6-phosphate-dehydrogenase), carrying the amino acid sequence NNWDN is considered as the precursor of EDF. An explanation is the degradation product of the G6PD which can be expected in conditions of very low nutrition is the EDF (You can easily see the point). MazE is degraded by the ATP-dependent ClpAP serine protease in the cell. By increasing the degradation of MazE the inhibitory effect of MazE is removed and MazF is free to perform its action. The MazE can also be reduced by stress related response such as DNA damage, Antibiotics such as rifampicins etc. Moreover, MazF activation led to an increase in the production of EDF. This is the positive feedback loop. Overall point is that when cell is required to suicide for the benefit of others (Altruistic behavior), such as in harsh conditions the cell can increase the activity of MazF and kill itself. I have presented an illustration of this idea in Fig 3.
This gives us a point of intervention. If we can design a chemical that can attack the antitoxin activity, the toxic activity of the MazF can be allowed to take over and kill the cell. The TAT system is usually very specific for a species, or a small group and hence specifically designed pharmacological compounds can be used, unlike the one's we use now which blindly attack many different bacteria including the normal flora. MazEF is not the only system in E coli. Other systems in E coli include chpBIK , relBE , yefM-yoeB and dinJ-yafQ systems. More and more studies are published in variety of bacteria showing similar systems, such as the most recently published PemIKSa system in S aureus.
That brings me to an important question. What about resistance. Say, I develop a compound that can interfere with this system, most probably by the law of natural selection since there will be a clear evolution. Antibiotic resistance can be expected in this case also.
Engelberg-Kulka H, Hazan R, & Amitai S (2005). mazEF: a chromosomal toxin-antitoxin module that triggers programmed cell death in bacteria. Journal of cell science, 118 (Pt 19), 4327-32 PMID: 16179604
Erental A, Sharon I, & Engelberg-Kulka H (2012). Two programmed cell death systems in Escherichia coli: an apoptotic-like death is inhibited by the mazEF-mediated death pathway. PLoS biology, 10 (3) PMID: 22412352
Bukowski M, Lyzen R, Helbin WM, Bonar E, Szalewska-Palasz A, Wegrzyn G, Dubin G, Dubin A, & Wladyka B (2013). A regulatory role for Staphylococcus aureus toxin-antitoxin system PemIKSa. Nature communications, 4 PMID: 23774061





Comments
Post a Comment