An Antibiotic that doesn't effect Gut Microbiome.
Greetings
Let us start with a question. What would be an ideal antibiotic? I can possibly talk about many different possibilities, but everyone would agree that we could boil it down to 2 most important points. First, the antibiotic needs to be as specific as possible and second absolutely no resistance. Though I have previously talked about these, it is important to summarise here once.
Antibiotics are usually targeted against a cellular component or enzymes of a vital biochemical pathway that kills the cell, in this case, bacteria. It is also necessary that this target exists in all clones of the target organism and forms vital and stable part of bacterial cell. In an evolutionary sense, such a component will also be present in other species, thus making it nearly impossible to design an antibiotic that will be hyper-specific to a species. When an antibiotic is made that is extremely species specific, chances of resistance is very high since the target can be lost easily without much evolutionary pressure on the bacterial cell. However, a closely balanced antibiotic design can in theory at least, be specific to a group of species.
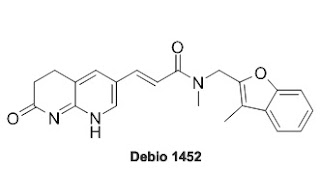 |
| Fig 1: Chemical Structure of Debio 1452. Source |
Researchers from St. Jude Children’s Research Hospital in Memphis, Tennessee, have found that a drug Debio 1452 can attack Staphylococcus aureus, with a very little impact on intestinal bacterial communities in mice. Debio 1452 has been in development since 2007 by Affinium Pharmaceuticals under the name AFN-1252. Debio 1452, is currently being developed by a Swiss-based pharmaceutical company Debiopharm. The chemical targets an enzyme called FabI (Eenoyl-acyl carrier protein reductase) in Staphylococcus species. Its lack of activity against other species of bacteria such as Streptococci, Enterococci, Enterobacteriaceae, and Non-fermentative Gram-negative species. This has allowed its development as a narrow spectrum drug. Fig 2, is a comparison of MIC aganist other well known anitiobitic for CoNS. I have made a graph for comparison.
 |
| Fig 2: Activity of Debio1452 in MIC50 (μg/ml) in comparison with antimicrobial agents against Coagulase-negative staphylococci. Data modified from Farrel etal. Source |
The ability of Debio 1452 to perform as an antibiotic has been previously tested. The aim of the current study was to see how far is the contribution of this drug in altering microbiome. For the study, mice in groups of five were treated with high doses of Debio 1452 and compared with four broad spectrum antibiotics every day for a period of 10 days. The gut microbiome was tracked using 16S ribosomal DNA sequencing on stool samples taken throughout the experiment and followed for 27 days. Analysis showed that Debio 1452-treated group had a very low variation of the microbiome.
It should be noted that the consistency of microbiome was not absolute. The treatment had a significant impact on a family of bacteria S24-7. The decrease in S24-7 was however quickly compensated for the imbalance by other members of microbiome and overall bacterial diversity and abundance wasn’t damaged during treatment. It took about two days post treatment for S24-7 levels to come back to its normal levels. As the authors point out in the abstract, "The gut bacterial abundance and composition of Debio 1452-treated mice was indistinguishable from untreated mice 2 days after antibiotic treatment stopped". It took much long time for mice treated with other broad-spectrum antibiotics to return to normal.
Yao J, Carter RA, Vuagniaux G, Barbier M, Rosch JW, & Rock CO (2016). A Pathogen-Selective Antibiotic Minimizes Disturbance to the Microbiome. Antimicrobial agents and chemotherapy PMID: 27161626





Comments
Post a Comment